Category: 2021
| Authors: |
Dries De Sloovere, Danny E. P. Vanpoucke, Andreas Paulus, Bjorn Joos, Lavinia Calvi, Thomas Vranken, Gunter Reekmans, Peter Adriaensens, Nicolas Eshraghi, Abdelfattah Mahmoud, Frédéric Boschini, Mohammadhosein Safari, Marlies K. Van Bael, An Hardy |
| Journal: |
Advanced Energy and Sustainability Research 3(3), 2100159 (2022) |
| doi: |
10.1002/aesr.202100159 |
| IF(2022): |
?? |
| export: |
bibtex |
| pdf: |
<AdvEnSusRes> (OA) |
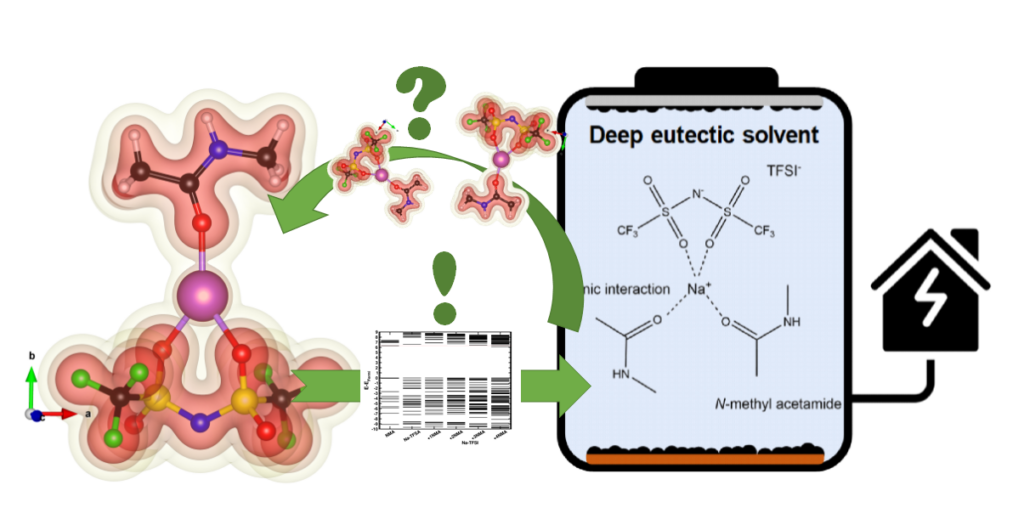 |
| Graphical Abstract: Understanding the electronic structure of Na-TFSI interacting with NMA. |
Sodium-ion batteries are alternatives for lithium-ion batteries in applications where cost-effectiveness is of primary concern, such as stationary energy storage. The stability of sodium-ion batteries is limited by the current generation of electrolytes, particularly at higher temperatures. Therefore, the search for an electrolyte which is stable at these temperatures is of utmost importance. Here, we introduce such an electrolyte using non-flammable deep eutectic solvents, consisting of sodium bis(trifluoromethane)sulfonimide (NaTFSI) dissolved in N-methyl acetamide (NMA). Increasing the NaTFSI concentration replaces NMA-NMA hydrogen bonds with strong ionic interactions between NMA, Na+, and TFSI–. These interactions lower NMA’s HOMO energy level compared to that of TFSI–, leading to an increased anodic stability (up to ~4.65 V vs Na+/Na). (Na3V2(PO4)2F3/CNT)/(Na2+xTi4O9/C) full cells show 74.8% capacity retention after 1000 cycles at 1 C and 55 °C, and 97.0% capacity retention after 250 cycles at 0.2 C and 55 °C. This is considerably higher than for (Na3V2(PO4)2F3/CNT)/(Na2+xTi4O9/C) full cells containing a conventional electrolyte. According to the electrochemical impedance analysis, the improved electrochemical stability is linked to the formation of more robust surface films at the electrode/electrolyte interface. The improved durability and safety highlight that deep eutectic solvents can be viable electrolyte alternatives for sodium-ion batteries.
Permanent link to this article: https://dannyvanpoucke.be/paper-desbatteries_dries-en/
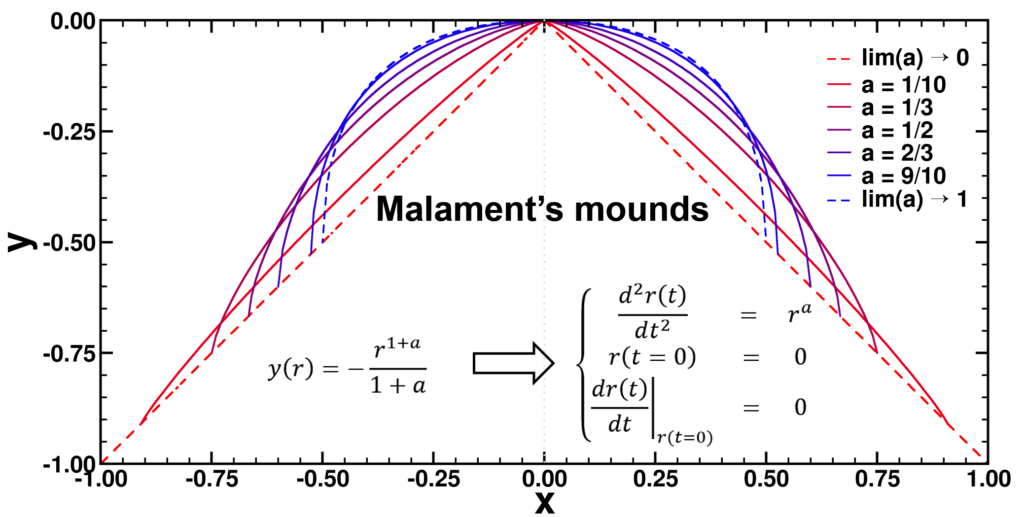 |
| Graphical Abstract: Examples of Malament’s Mounds, of which the Norton Dome is the special case where α=½. |
We present a method for assigning probabilities to the solutions of initial value problems that have a Lipschitz singularity. To illustrate the method, we focus on the following toy-example:  ,
, 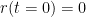 , and
, and  , with a∈]0; 1[. This example has a physical interpretation as a mass in a uniform gravitational field on a dome of particular shape; the case with a = ½ is known as Norton’s dome. Our approach is based on (1) finite difference equations, which are deterministic; (2) elementary techniques from alpha-theory, a simplified framework for non-standard analysis that allows us to study infinitesimal perturbations; and (3) a uniform prior on the canonical phase space. Our deterministic, hyperfinite grid model allows us to assign probabilities to the solutions of the This allows us to assign probabilities to the solutions of the initial value problem in the original, indeterministic model.
, with a∈]0; 1[. This example has a physical interpretation as a mass in a uniform gravitational field on a dome of particular shape; the case with a = ½ is known as Norton’s dome. Our approach is based on (1) finite difference equations, which are deterministic; (2) elementary techniques from alpha-theory, a simplified framework for non-standard analysis that allows us to study infinitesimal perturbations; and (3) a uniform prior on the canonical phase space. Our deterministic, hyperfinite grid model allows us to assign probabilities to the solutions of the This allows us to assign probabilities to the solutions of the initial value problem in the original, indeterministic model.
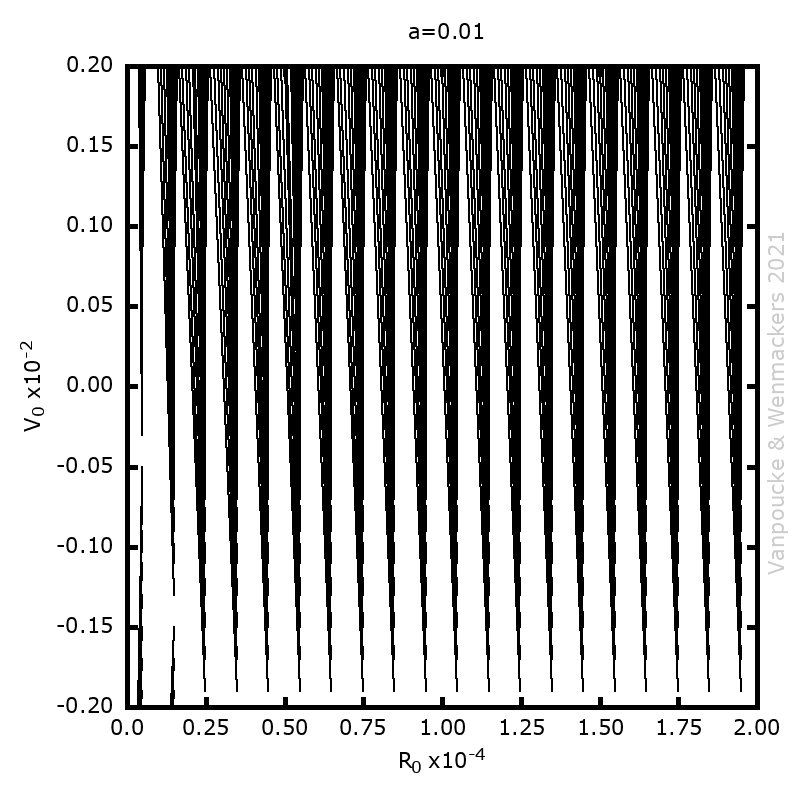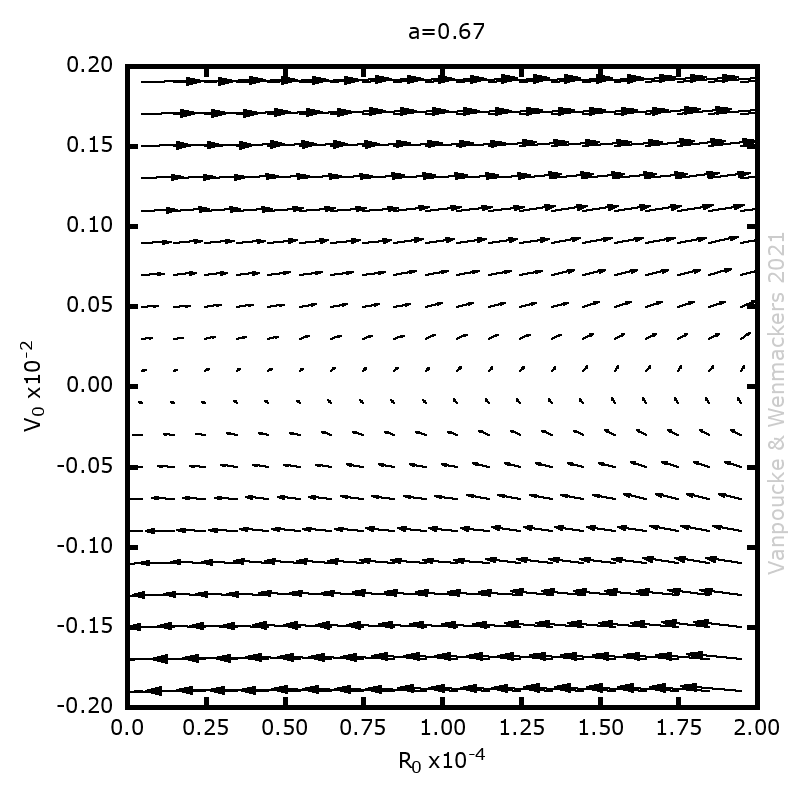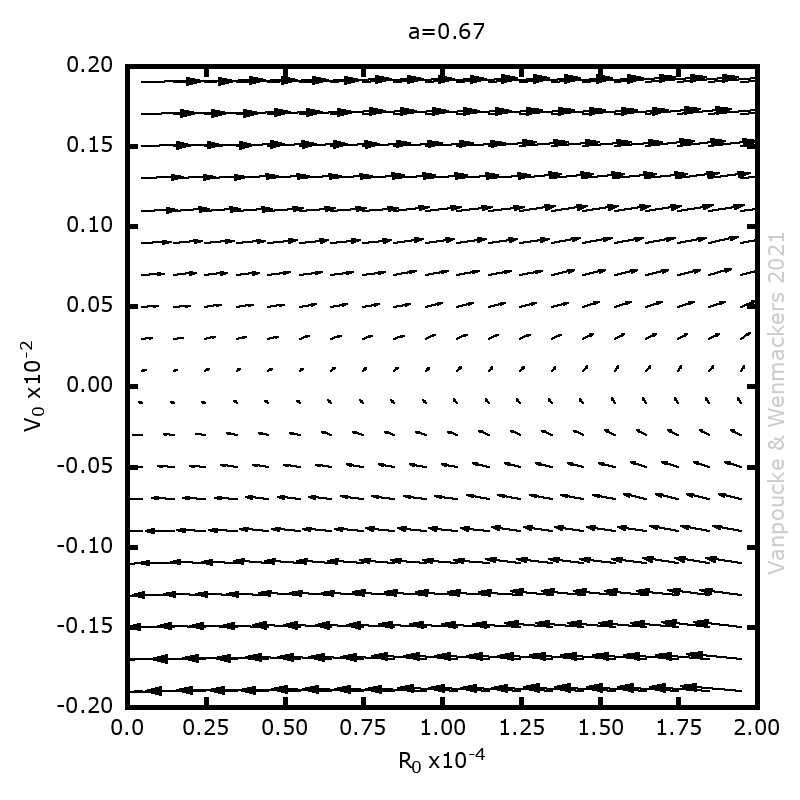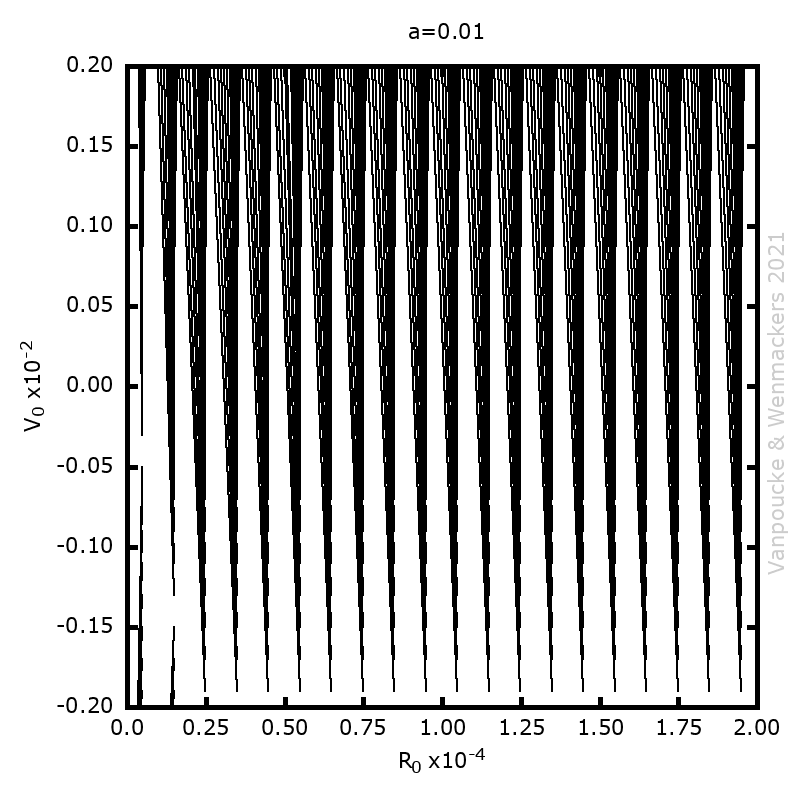
Phase space vector field for all Malament’s mounds
Permanent link to this article: https://dannyvanpoucke.be/paper_domeprogram-en/
Happy New Year
2020 will forever be the year of viruses for me and a lot of us. At Maastricht University, the year started with a university wide cyber-attack with ransomware. After the computer-viruses came the human viruses, with COVID-19 shutting down one country after the other, and shutting down education systems as well.
Hopefully 2021 will be better behaved, though we know already some of the hurdles which will make life interesting the coming year. COVID-19 is far from over, and it will take at least a year to vaccinate everyone. Furthermore, as of the first of today, the United Kingdom is no longer a part of the EU, making travel inside Europe a little harder again.
But before we launch into these new and interesting times, lets look back at 2020 one last time, keeping up with tradition. What have I done during the last year of academic merit.
1. Publications: +6 (and currently a handful in progress)
-
Rozita Rouzbahani, Shannon S. Nicley, Danny E. P. Vanpoucke, Fernando Lloret, Paulius Pobendinskas, Daniel Araujo, and Ken Haenen,
-
Danny E. P. Vanpoucke, Onno S. J. van Knippenberg, Ko Hermans, Katrien V. Bernaerts, and Siamak Mehrkanoon
-
-
Viraj Damle, Kaiqi Wu, Oreste De Luca, Natalia Ortí-Casañ, Neda Norouzi, Aryan Morita, Joop de Vries, Hans Kaper, Inge Zuhorn, Ulrich Eisel, Danny E.P. Vanpoucke, Petra Rudolf, and Romana Schirhagl,
-
Jules Stouten, Danny E. P. Vanpoucke, Guy Van Assche, and Katrien V. Bernaerts,
-
Mohammadreza Hosseini, Danny E.P. Vanpoucke, Paolo Giannozzi, Masoud Berahman, Nasser Hadipour,
2. Completed refereeing tasks: +17
- Applied Physics Letters
- Journal of Physical Chemistry (2x)
- Computational Materials Science (2x)
- Materials Chemistry and Physics
- Journal of Physics: Condensed Matter (5x)
- Diamond and Related Materials (6x)
3. Conferences & workshops in times of Corona: +3/+1 (Attended & Organised), >+4 internal

ACOS poster prize 2020
With regard to conferences, 2020 was the year everyone came into contact with the concept of the online conference. Many conferences and events got canceled: such as TEDx@UHasselt (which will return in 2021)
- ACOS 2020, Online, Oktober 28th, 2020 [poster presentation and video-pitch, 2nd poster prize]
- RSC Chemical Science Symposium 2020, Online, September 29th-30th, 2020 [iposter presentation]
- D-NL-HIT project meetings [oral presentations]
- Virtual Partner Meeting, April 8th, 2020
- Adhesives Pilot Branch meeting, October 7th, 2020
- Virtual Partner Meeting, October 15th, 2020
- UV-Curing Branch meeting, October 22nd, 2020
- SBDD XXV, Hasselt University, Belgium, March 11th-13th, 2020 [(invited) oral presentation, poster presentation] …On Friday13th Belgium went into it’s first lock-down.
- Pilot Branch meeting adhesives D-NL-HIT project, Maastricht University, Brighlands campus, February 26th, 2020 [Organised]
4. Science Communication & Social media:
- In February 2020, I finally caved and joined Twitter as @DelocalizedD .
- Added several new repositories to my github account, with the most important ones being:
- Started a YouTube channel (for the ACOS video pitch)
5. Current size of HIVE:
- Continued work on a public version of HIVE at github: HIVE 4.x (26K lines, 9 commands available)
- 61K lines of program (code: 69 %)
- ~100 files
- 49 (command line) options
6. Hive-STM program:
And now, upward and onward, a new year, a fresh start.
Permanent link to this article: https://dannyvanpoucke.be/review-of-2020/
| Authors: |
Rozita Rouzbahani, Shannon S.Nicley, Danny E.P.Vanpoucke, Fernando Lloret, Paulius Pobedinskas, Daniel Araujo, Ken Haenen |
| Journal: |
Carbon 172, 463-473 (2021) |
| doi: |
10.1016/j.carbon.2020.10.061 |
| IF(2019): |
8.821 |
| export: |
bibtex |
| pdf: |
<Carbon> |
 |
| Graphical Abstract: Artist impression of B incorporation during CVD growth of diamond. |
The methane concentration dependence of the plasma gas phase on surface morphology and boron incorporation in single crystal, boron-doped diamond deposition is experimentally and computationally investigated. Starting at 1%, an increase of the methane concentration results in an observable increase of the B-doping level up to 1.7×1021 cm−3, while the hole Hall carrier mobility decreases to 0.7±0.2 cm2 V−1 s−1. For B-doped SCD films grown at 1%, 2%, and 3% [CH4]/[H2], the electrical conductivity and mobility show no temperature-dependent behavior due to the metallic-like conduction mechanism occurring beyond the Mott transition. First principles calculations are used to investigate the origin of the increased boron incorporation. While the increased formation of growth centers directly related to the methane concentration does not significantly change the adsorption energy of boron at nearby sites, they dramatically increase the formation of missing H defects acting as preferential boron incorporation sites, indirectly increasing the boron incorporation. This not only indicates that the optimized methane concentration possesses a large potential for controlling the boron concentration levels in the diamond, but also enables optimization of the growth morphology. The calculations provide a route to understand impurity incorporation in diamond on a general level, of great importance for color center formation.
Permanent link to this article: https://dannyvanpoucke.be/paper_bdoping-en/






